what quantum numbers specify these subshells?
Web See Answer What quantum numbers specify these subshells. 3s 4p 3d May I please get steps and explanations.
 |
| Solved How To Identify Which Set Of Quantum Numbers Contains Error Each Course Hero |
7s 5p 3d n n n l l l Expert Answer 100 28 ratings Previous question Next question.
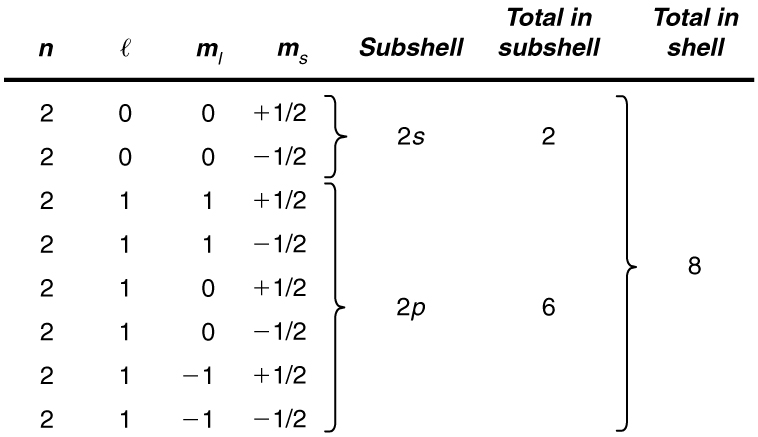
. Web See Answer What quantum numbers specify these subshells. 4s n l 5p n l 3d n l This problem has been solved. Web What quantum numbers specify these subshells. Principal quantum number n.
Web Question What quantum numbers specify these subshells. Web The azimuthal quantum number l defines the subshell of the orbital. Web What quantum numbers specify these subshells. Youll get a detailed solution from a subject matter expert that helps you learn.
What quantum numbers specify these subshells. 3s 2p 6d This problem has been solved. I am very confused with quantum numbers. In order these four quantum numbers are.
ℓ 7s n ℓ 2p n ℓ 6d n ℓ Follow 1 Add comment Report 1 Expert Answer Best Newest Oldest Luv A. Given - subshells. For 4p orbital the value of l can be 0 1 2 3. Web What quantum numbers specify these subshells 4s 5p 3d Follow 2 Add comment Report 2 Answers By Expert Tutors Best Newest Oldest JR.
A 2S B 2P and C 6D. It is calculated as the difference between the n and 1. So the principal quantum number n for the 5p. 6s n l 4p n l 5d n l.
1s 5p 3d to be determined - their n and l values. Each set is of the form. Ill start with the first. Web As Im sure you know there are four quantum numbers used to describe the location of an electron.
5 1 -1 -12 5 1 0 -12 5 1 1 -12 Give. ℓ Expert Solution Want to see the full answer. Check out a sample QA here See Solution. Need the values of n and l.
Given - subshells. Web What quantum numbers specify these subshells. What quantum numbers specify these subshells. 4𝑠 𝑛 ℓ 5𝑝 𝑛 ℓ 6𝑑 𝑛 ℓ Expert Answer 100 6 ratings Principal Quantum Number n - Principal Quantum Number.
What quantum numbers specify these subshells. Web Classify each set of quantum numbers as valid or invalid for an electron in a 5p orbital. QA By tamdoan June 23 2021 0 Comment 2 Answers 6s n is 6 L is 0 2p n is 2 L is. Youll get a detailed solution from a subject matter expert.
1s 5p 3d to be determined - their n and l values. N l ml ms. What quantum numbers specify these subshells. Best Answer 100 4 ratings s0p1d2f3 and n is just the first.
1s 3D n 5p n 3d. Web See the answer What quantum numbers specify these subshells. Web In your case the energy level is given by the number thats placed in front of the letter p which denotes a specific subshell.
 |
| Organization Of Electrons In Atoms Introductory Chemistry 1st Canadian Edition |
 |
| Magnetic Quantum Number Examples What Are Quantum Numbers In Chemistry Video Lesson Transcript Study Com |
 |
| How To Calculate The Number Of Electrons In Mn Having A Magnetic Quantum Number 0 Quora |
 |
| What Are The Possible Value For N L M And S For 3s Electron Quora |
 |
| What Are Quantum Numbers And What Information Is Provided By Them Specify The Electrons With Following Sets Of Quantum Numbers I N 4 L 1 M 1 S 1 2 Ii N 3 L 0 M 0 S 1 2 |
Posting Komentar untuk "what quantum numbers specify these subshells?"